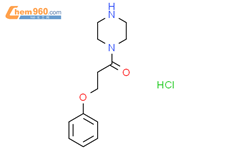
1.1
Reagents:
Potassium tert-butoxide
Catalysts:
Copper sulfate
Solvents:
1,4-Dioxane
;
24 h, 90 °C
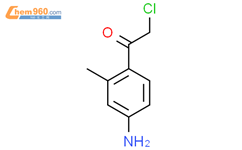
1.2
Reagents:
Trifluoroacetic acid
,
2-Mercaptobenzoic acid
;
12 h, 30 °C
找化学品上960化工网!



 2881489226
2881489226 












































![(1R,4R)-2,5-二苯基二环[2.2.2]辛-2,5-二烯 (1R,4R)-2,5-二苯基二环[2.2.2]辛-2,5-二烯](http://hg.y866.cn/compound/lib/scimg/cas/150/796966-15-9.png)