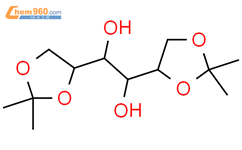
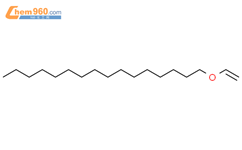
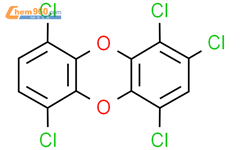
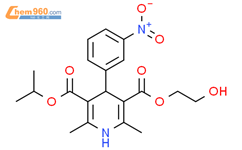
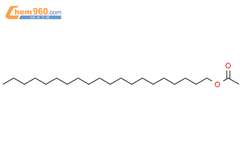
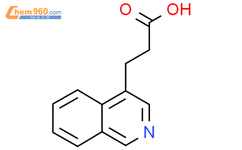
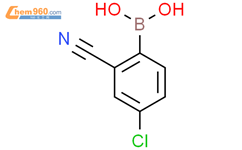
1.1
Reagents:
Sodium bicarbonate
Solvents:
Tetrahydrofuran
,
Water
;
rt → 0 °C; 1 h, 0 °C
1.2
Reagents:
Hydrochloric acid
Solvents:
Water
;
0 °C


 2881489226
2881489226 















![3-(2-对甲苯基咪唑[1,2-a]-3-吡啶基)丙烯酸 3-(2-对甲苯基咪唑[1,2-a]-3-吡啶基)丙烯酸](http://hg.y866.cn/compound/lib/scimg/cas/150/817172-45-5.png)