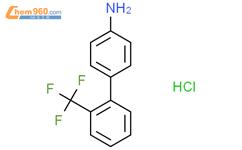
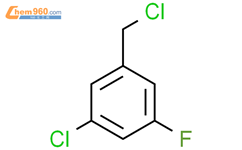
1.1
Reagents:
1-Hydroxybenzotriazole
,
Diisopropylethylamine
,
1-[Bis(dimethylamino)methylene]-1H-benzotriazolium hexafluorophosphate(1-) 3-oxi…
Solvents:
Dimethylformamide
;
1 - 3 h, rt
1.2
Reagents:
Piperidine
Solvents:
Dimethylformamide
;
rt
1.3
Reagents:
1-Hydroxybenzotriazole
,
Diisopropylethylamine
,
1-[Bis(dimethylamino)methylene]-1H-benzotriazolium hexafluorophosphate(1-) 3-oxi…
Solvents:
Dimethylformamide
;
1 - 3 h, rt
1.4
Reagents:
Piperidine
Solvents:
Dimethylformamide
;
rt
1.5
Reagents:
1-Hydroxybenzotriazole
,
Diisopropylethylamine
,
1-[Bis(dimethylamino)methylene]-1H-benzotriazolium hexafluorophosphate(1-) 3-oxi…
Solvents:
Dimethylformamide
;
1 - 3 h, rt
1.6
Reagents:
Piperidine
Solvents:
Dimethylformamide
;
rt
1.7
Reagents:
Acetic acid
,
2,2,2-Trifluoroethanol
Solvents:
Dichloromethane
;
90 min, rt
1.8
Reagents:
Sodium bicarbonate
,
Diphenylphosphoryl azide
Solvents:
Dimethylformamide
;
48 h, rt
1.9
Reagents:
Trifluoroacetic acid
Solvents:
Water
;
rt
找化学品上960化工网!



 764619768
764619768 




































![5'-O-[二(4-甲氧基苯基)苯基甲基]-3'-O-[(叔丁基)二甲基硅烷基]尿苷 5'-O-[二(4-甲氧基苯基)苯基甲基]-3'-O-[(叔丁基)二甲基硅烷基]尿苷](http://hg.y866.cn/compound/lib/scimg/cas/150/81246-81-3.png)