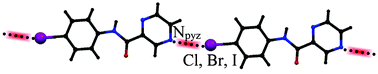
A series of halogen-substituted phenylpyrazinamides have been synthesized and crystallographically characterized in order to investigate the effect of halogen bonding interaction on supramolecular assembly of N-phenylpyrazine-2-carboxamide derivatives. The notable feature in crystal structures of meta- and para-iodinated, brominated and chlorinated compounds is that there is a tendency to form a halogen bonding synthon between adjacent halophenyl and prazine/halophenyl rings. Influence of these halogen bonding interactions on supramolecular assemblies have been discussed with the help of geometrical analysis and theoretical calculations. The X⋯N halogen bonding distances are 2.2–7.7% shorter than the sum of the van der Waals radii of the nitrogen and halogen atoms. Also, theoretical methods show the N⋯X halogen bonding energies within a range of −9.43 to −23.67 kJ mol−1. Our studies show that the selection of halogen atom as well as the position of substitution on phenylpyrazinamide compound may be important for crystal design based on halogen bonding.