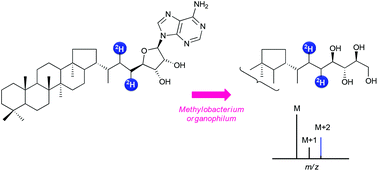
Adenosylhopane is a putative precursor of the widespread bacterial C35 biohopanoids. A concise and flexible hemisynthesis of adenosylhopane has been developed including as key steps a cross metathesis between two olefins containing either the hopane moiety or a protected adenosine derivative and a subsequent diimide reduction of the resulting olefin. Reduction by deuteriated diimide allowed deuterium labelling. This synthetic protocol represents a versatile tool to access to deuteriated composite bacterial hopanoids required for biosynthetic studies. Deuteriated adenosylhopane was thus converted into bacteriohopanetetrol by a crude cell-free system from Methylobacterium organophilum in the presence of NADPH, showing for the first time the precursor to product relationship between these two bacterial metabolites.