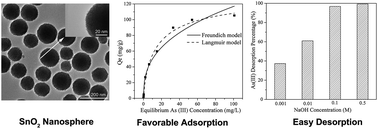
The development of highly efficient As(III) adsorbents is critical to largely simplify the arsenic treatment process and lower its cost. For the first time, SnO2 nanospheres were demonstrated to possess a highly efficient As(III) adsorption capability from water in a near neutral pH environment as predicted by the material criterion we recently developed for the selection of highly efficient arsenic adsorbents. These SnO2 nanospheres were synthesized by a simple and cost-effective hydrolysis process with the assistance of ethyl acetate under ambient conditions, which had a good dispersity, a narrow size distribution, a relatively large specific surface area, and a porous structure. A fast As(III) adsorption was observed in the kinetics study on these SnO2 nanospheres, and their Langmuir adsorption capacity was determined to be ∼112.7 mg g−1 at pH ∼7. The As(III) adsorption mechanism on SnO2 nanospheres was examined by both macroscopic and microscopic techniques, which demonstrated that it followed the inner-sphere complex model. These SnO2 nanospheres demonstrated effective As(III) adsorption even with exceptionally high concentrations of co-existing ions, and a good regeneration capability by washing with NaOH solution.