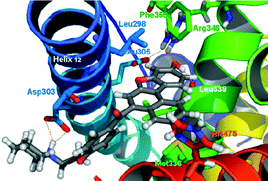
Based on the benzo[b]naphtho[1,2-d]furan and benzo[b]naphtho[1,2-d]thiophene frameworks, a series of ligands with different basic side chains (BSCs) has been synthesized and pharmacologically evaluated. Also, their binding modes have been modelled using docking techniques. It was found that the introduction of a BSC in these systems brings about a decrease of affinity for both estrogen receptors α and β in an in vitro competitive binding assay. However, two full antagonists of the estrogen receptor β (9c and 9f) have been discovered, with potency in the low micromolar concentration in a cell-based luciferase reporter assay, and completely devoid of activity against the α receptor at the same concentration range. Differences in the ERα/ERβ binding modes have also been rationalized with the help of molecular modelling techniques. This interesting functional profile could be used to elucidate the physiological role of each ER subtype.