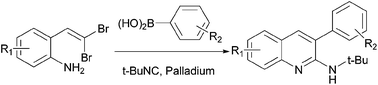
A three-component cascade reaction involving gem-dibromovinylanilines, tert-butyl isocyanide and arylboronic acids for the efficient synthesis of 3-aryl-2-aminoquinolines has been developed. The reaction proceeds through palladium-catalyzed isocyanide insertion, intramolecular cyclization of gem-dibromovinylanilines followed by Suzuki coupling with arylboronic acids, and the corresponding products were obtained in good to excellent isolated yields.