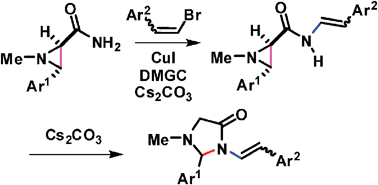
N-Styryl-3-aryl-1-methylaziridine-2-carboxamides, which were readily obtained from the cross coupling reaction between 3-aryl-1-methylaziridine-2-carboxamides and 1-aryl-2-bromoethenes catalyzed by CuI/N,N-dimethylglycine in the presence of Cs2CO3, underwent a base-mediated intramolecular nucleophilic aziridine ring opening reaction effectively via the carbon–carbon bond cleavage of aziridine to afford the ring expanded imidazolidin-4-one products in good yields.