找化学品上960化工网!
We present a method for approximating thermodynamic parameters 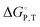 , ΔH, and ΔS for the cation exchange process in metal–organic frameworks, as exemplified by Ni2+ exchange into Zn4O(1,4-benzenedicarboxylate)3 (MOF-5) and Co2+ exchange into MOF-5 and Zn5Cl4(bis(1H-1,2,3-triazolo-[4,5-b],[4′,5′-i])dibenzo-[1,4]-dioxin)3 (MFU-4l). For these examples, we find that the cation exchange process is endergonic and that parameters such as solvent and cation identity impact the thermodynamics.
, ΔH, and ΔS for the cation exchange process in metal–organic frameworks, as exemplified by Ni2+ exchange into Zn4O(1,4-benzenedicarboxylate)3 (MOF-5) and Co2+ exchange into MOF-5 and Zn5Cl4(bis(1H-1,2,3-triazolo-[4,5-b],[4′,5′-i])dibenzo-[1,4]-dioxin)3 (MFU-4l). For these examples, we find that the cation exchange process is endergonic and that parameters such as solvent and cation identity impact the thermodynamics.
