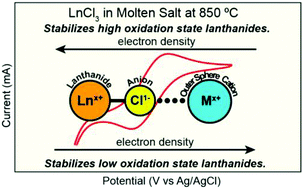
Controlling structure and reactivity by manipulating the outer-coordination sphere around a given reagent represents a longstanding challenge in chemistry. Despite advances toward solving this problem, it remains difficult to experimentally interrogate and characterize outer-coordination sphere impact. This work describes an alternative approach that quantifies outer-coordination sphere effects. It shows how molten salt metal chlorides (MCln; M = K, Na, n = 1; M = Ca, n = 2) provided excellent platforms for experimentally characterizing the influence of the outer-coordination sphere cations (Mn+) on redox reactions accessible to lanthanide ions; Ln3+ + e1− → Ln2+ (Ln = Eu, Yb, Sm; e1− = electron). As a representative example, X-ray absorption spectroscopy and cyclic voltammetry results showed that Eu2+ instantaneously formed when Eu3+ dissolved in molten chloride salts that had strongly polarizing cations (like Ca2+ from CaCl2) via the Eu3+ + Cl1− → Eu2+ + ½Cl2 reaction. Conversely, molten salts with less polarizing outer-sphere M1+ cations (e.g., K1+ in KCl) stabilized Ln3+. For instance, the Eu3+/Eu2+ reduction potential was >0.5 V more positive in CaCl2 than in KCl. In accordance with first-principle molecular dynamics (FPMD) simulations, we postulated that hard Mn+ cations (high polarization power) inductively removed electron density from Lnn+ across Ln–Cl⋯Mn+ networks and stabilized electron-rich and low oxidation state Ln2+ ions. Conversely, less polarizing Mn+ cations (like K1+) left electron density on Lnn+ and stabilized electron-deficient and high-oxidation state Ln3+ ions.