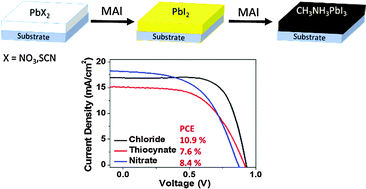
We report the formation of CH3NH3PbI3 from more soluble, non-iodide lead salts like Pb(SCN)2 and Pb(NO3)2. When exposed to CH3NH3I vapours, the colourless lead salts turned yellow before the formation of the black perovskite. Investigation of this yellow intermediate suggests that anion exchange (converting lead salts to PbI2) precedes the perovskite formation. PCEs of 7.6% and 8.4% were achieved for the devices formed from Pb(SCN)2 and Pb(NO3)2, respectively.