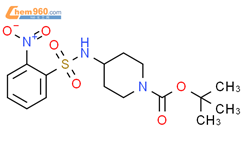
1.1
Reagents:
Oxygen
Catalysts:
1-Methylimidazole
,
4,4′-Dimethoxy-2,2′-bipyridine
,
9-Azabicyclo[3.3.1]non-9-yloxy
,
Copper(I) triflate
Solvents:
Acetonitrile
;
1 min, 24 atm, 100 °C
1.2
Reagents:
Ethyl acetate


 3001086693
3001086693 


































![5-bromo-1H-pyrrolo[3,2-b]pyridine-2-carboxylic acid 5-bromo-1H-pyrrolo[3,2-b]pyridine-2-carboxylic acid](http://hg.y866.cn/compound/lib/scimg/cas/150/800401-52-9.png)