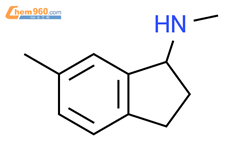
1.1
Reagents:
Sodium hydride
Solvents:
Dimethyl sulfoxide
,
Tetrahydrofuran
;
30 min, rt
1.2
Solvents:
Tetrahydrofuran
;
2 h, 0 °C; rt
1.3
Reagents:
Ammonium chloride
Solvents:
Water
;
rt



 3003949364
3003949364