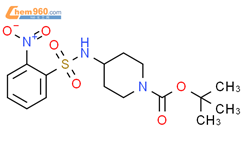
累匹利洛(91374-21-9)名称与标识符
名称
中文别名:
累匹利洛;罗匹尼罗;4-[2-(二丙胺基)乙基]-1,3-二氢吲哚-2-酮;罗匹尼罗 技术转让;罗匹尼罗-D7盐酸;4-[2-(二丙氨基)乙基]-1,3-二氢;4-[2-(二丙氨基)乙基]-1,3-二氢-2H-吲哚-2-酮;
英文别名:
Ropinirole;(Ropinirole );2H-Indol-2-one, 4-[2-(dipropylamino)ethyl]-1,3-dihydro-;4-(2-Dipropylaminoethyl)-1,3-dihydroindol-2-one;Ropinirole (as hydrochloride);ROPINIROLE INTERMEIDATES:;Ropinirolum;Unii-030pyr8953;4-[2-(Dipropylamino)ethyl]-1,3-dihydro-2H-indol-2-one (ACI);NP 201;SKF 101468;4-(2-(Dipropylamino)ethyl)indolin-2-one;NCGC00015893-03;AB01563044_02;BRD-K15933101-003-07-9;Ropinirole (USAN/INN);CS-0009561;2H-Indol-2-one, 4-(2-(dipropylamino)ethyl)-1,3-dihydro-;4-(2-(di-n-propylamino)ethyl)-2(3H)-indolone;Ropinirol (INN-Spanish);SKF-101468;GTPL7295;Lopac0_001101;NCGC00015893-04;4-[2-(dipropylamino)ethyl]-3H-indol-2-ol;G78293;Tox21_110256_1;ROPINIROLE [MI];Narapin;4-[2-(Dipropylamino)ethyl]2-indolinone;NCGC00015893-01;BRD-K15933101-003-01-2;SR-01000076215-3;91374-21-9;BCP09383;ROPINIROLE [USAN];L000520;Q420590;NCGC00096064-02;Ropitor (TN);CAS-91374-21-9;Lopac-R-4152;DTXSID8045195;N04BC04;CHEBI:8888;NSC758917;NCGC00096064-01;EN300-708794;Ropitor;4-[2-(dipropylamino)ethyl]-1,3-dihydro-2H-indol-2-one;NCGC00015893-02;SCHEMBL35212;ROPINIROLE [VANDF];030PYR8953;SDCCGSBI-0051070.P002;NCGC00094373-12;DB00268;AB01563044_01;BIDD:GT0826;ropinirol;4-(2-(Dipropylamino)ethyl)-1,3-dihydro-2H-indol-2-one;AKOS015843123;HMS2093K04;ROPINIROLE [INN];4-[2-(dipropylamino)ethyl]-1,3-dihydroindol-2-one;Ropinirol [INN-Spanish];Tox21_110256;C07564;4-[2-(dipropylamino)ethyl]-2,3-dihydro-1h-indol-2-one;CHEMBL589;NSC 758917;Ropinirolum (Latin);Ropinirolum (INN-Latin);CCG-205177;Ropinirolum [INN-Latin];BDBM50020680;NS00008049;HY-B0623;SB65618;SK&F 101468;HSDB 8252;D08489;Pharmakon1600-01505178;NSC-758917;Ropinirole [USAN:INN:BAN];BRD-K15933101-003-06-1;NCGC00015893-06;ROPINIROLE [WHO-DD];STL454344;SPECTRUM1505178;DTXCID6025195;4-[2-(dipropylamino)ethyl]-1.3-dihydro-2H-indol-2-one;SK&F-101468;
标识符
InChIKey:
UHSKFQJFRQCDBE-UHFFFAOYSA-N
Inchi:
1S/C16H24N2O/c1-3-9-18(10-4-2)11-8-13-6-5-7-15-14(13)12-16(19)17-15/h5-7H,3-4,8-12H2,1-2H3,(H,17,19)
SMILES:
O=C1CC2C(=CC=CC=2CCN(CCC)CCC)N1


 1400878899
1400878899 





















![(9Ci)-5,7-二氢-4,8-二甲基-苯并[1,2-B:4,5-B]二吡咯-2,6(1H,3H)-二酮 (9Ci)-5,7-二氢-4,8-二甲基-苯并[1,2-B:4,5-B]二吡咯-2,6(1H,3H)-二酮](http://hg.y866.cn/compound/lib/scimg/cas/150/397283-37-3.png)


![5-bromo-1H-pyrrolo[3,2-b]pyridine-2-carboxylic acid 5-bromo-1H-pyrrolo[3,2-b]pyridine-2-carboxylic acid](http://hg.y866.cn/compound/lib/scimg/cas/150/800401-52-9.png)