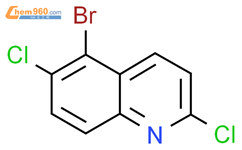
1.1
Reagents:
N-Bromosuccinimide
,
Sodium hydroxide
Solvents:
Water
;
rt; 1 h, rt
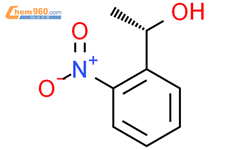
1.2
Reagents:
Hydrochloric acid
Solvents:
Water
;
pH 2 - 3


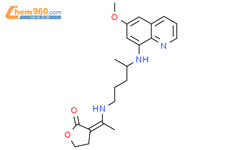
 2881489226
2881489226 
























![(2S)-2-({[(1R,7aR)-1-(乙酰氧基)-2,3,5,7a-四氢-1H-吡咯嗪-7-基]甲氧基}羰基)-2-羟基-1,3-二甲基丁基(2E)-2-甲基丁-2-烯酸酯 (2S)-2-({[(1R,7aR)-1-(乙酰氧基)-2,3,5,7a-四氢-1H-吡咯嗪-7-基]甲氧基}羰基)-2-羟基-1,3-二甲基丁基(2E)-2-甲基丁-2-烯酸酯](http://hg.y866.cn/compound/lib/scimg/cas/150/80405-17-0.png)
![(1S,2S)-2-羟基-2-({[(1R,7aR)-1-羟基-2,3,5,7a-四氢-1H-吡咯嗪-7-基]甲氧基}羰基)-1,3-二甲基丁基(2E)-2-甲基丁-2-烯酸酯 (1S,2S)-2-羟基-2-({[(1R,7aR)-1-羟基-2,3,5,7a-四氢-1H-吡咯嗪-7-基]甲氧基}羰基)-1,3-二甲基丁基(2E)-2-甲基丁-2-烯酸酯](http://hg.y866.cn/compound/lib/scimg/cas/150/80405-18-1.png)